foodies
Health & Food Tips by Calenos Jola
Editor’s Pick
articles for you
Amanda Furness
September 28, 2022
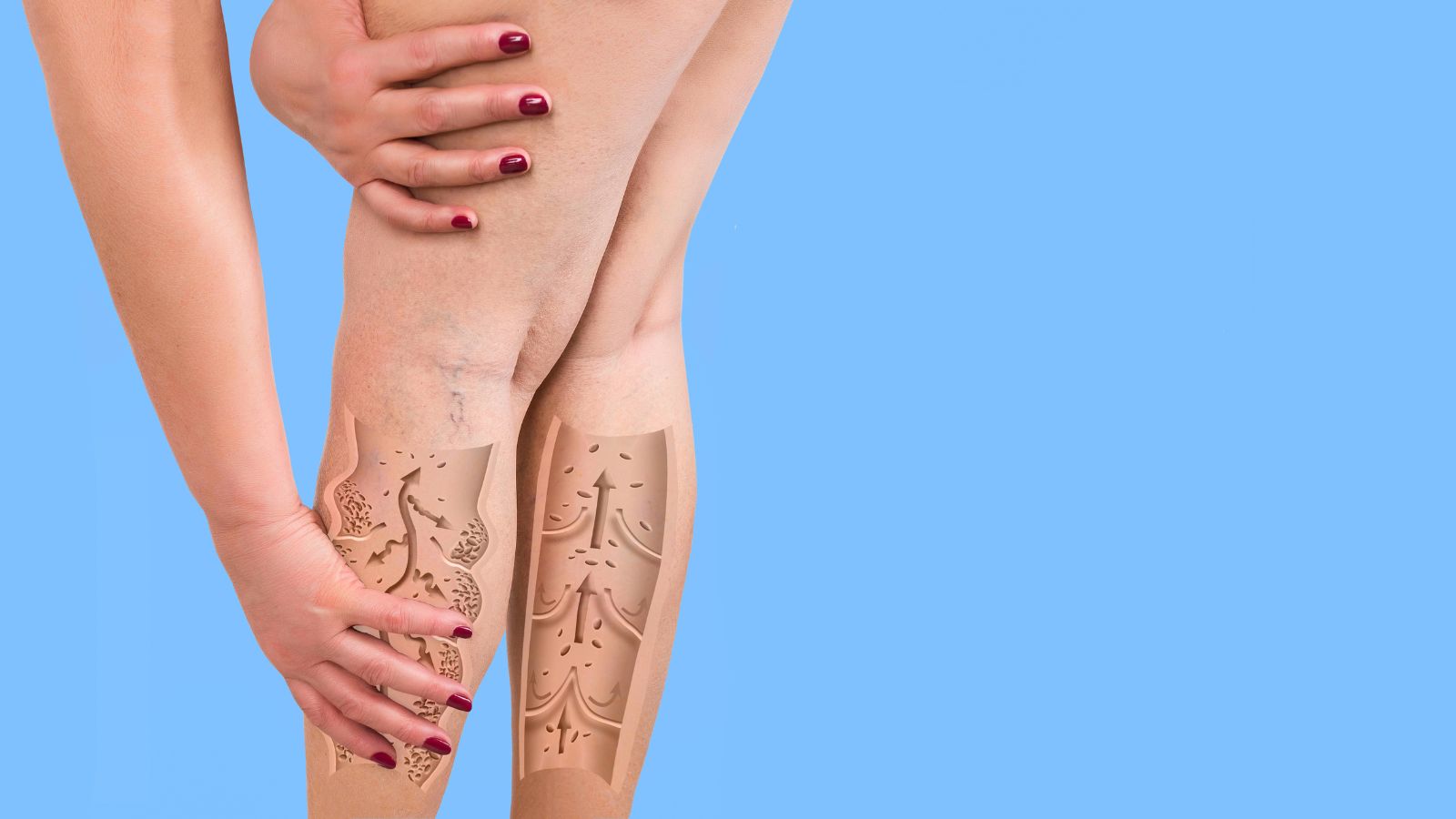
Twitter is a great way to stay connected with friends and family, but it’s important to remember...
Amanda Furness
September 27, 2022
Muting Other Sounds on Windows 10: Microsoft, in their infinite wisdom and cack-handedness (delete as appropriate) have...
Calenos Jola
December 24, 2021
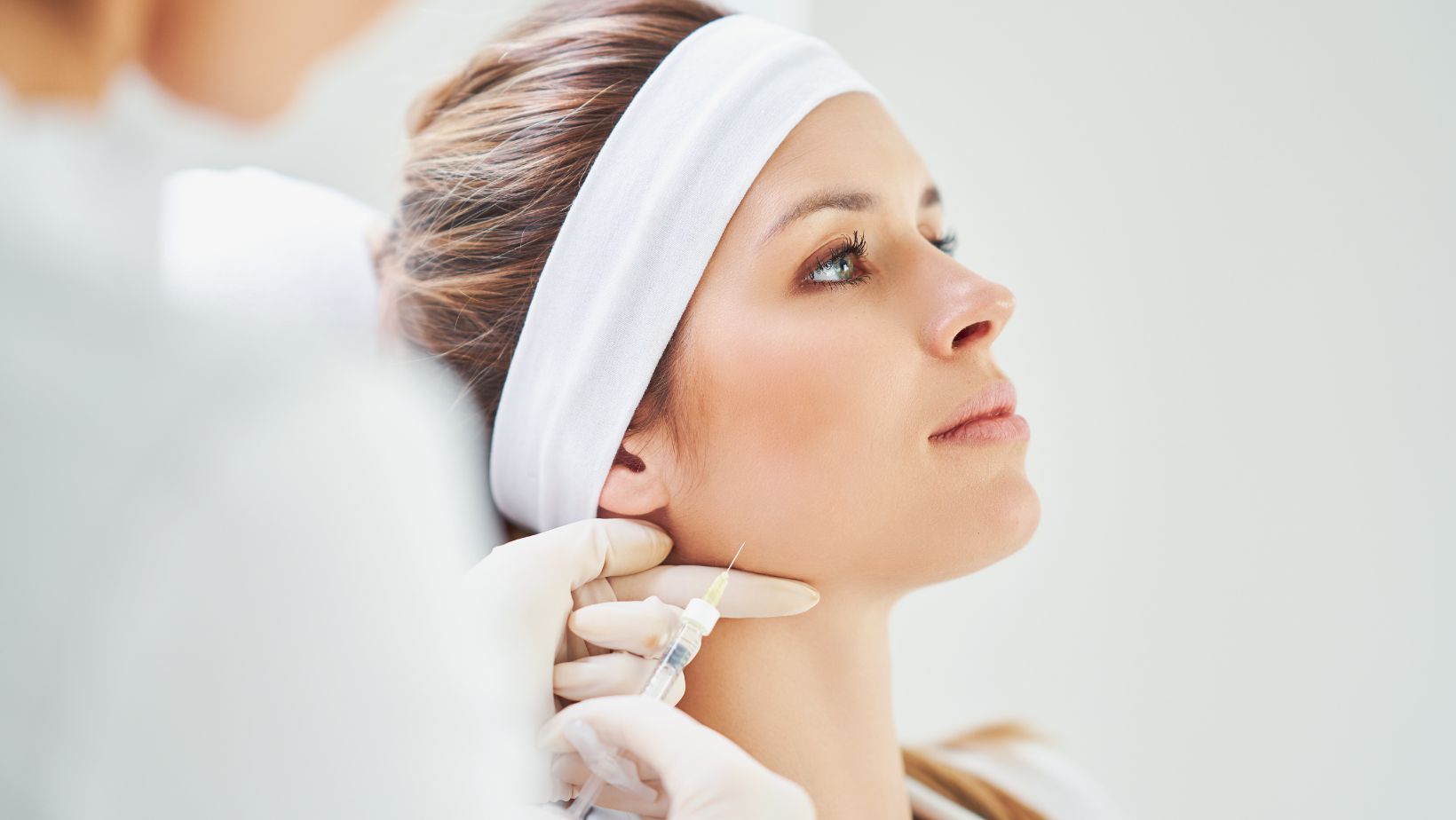
The AI-powered DreamStation mask has been designed to make it easier for patients to use the device...
Amanda Furness
December 10, 2021
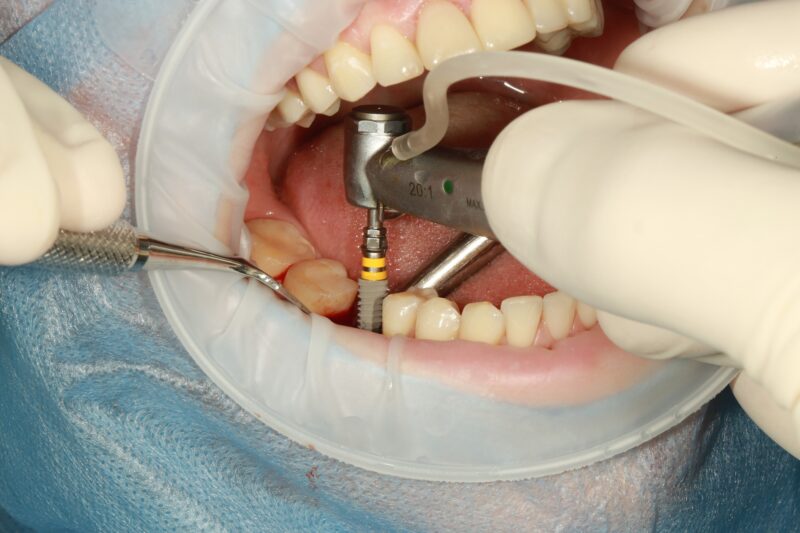
Dental implants are more popular than ever, with about 10 million people in the U.S. receiving them...